Let's get this out of the way first: Benchling is a good product. It has a clean interface, strong molecular biology tools, and it's become something of a default choice in biotech - especially at companies managed by people that started using the free academic tier in grad school and carried that familiarity into industry. If Benchling is working well for your lab, there's no reason to switch.
But increasingly, labs are looking for Benchling alternatives, naming pricing as a significant factor. Startup plans are reported to start around $15,000 per year, with enterprise contracts scaling well beyond that - and because Benchling doesn't publish pricing publicly, true cost comparison is difficult until you're already deep in a sales conversation. What often catches labs off guard is the sharp price increase that kicks in once a team outgrows the team-size limit for the startup pricing tier. We've spoken to many teams who reached that point and found they could no longer justify the cost.
Then there's the learning curve. Benchling is a complex piece of software, and reviewers consistently flag that it's not particularly intuitive for first-time users. There's also what often becomes an invisible headcount cost: in practice, many labs end up with someone functioning as a de facto internal Benchling administrator, spending a significant portion of their time on configurations, onboarding, and troubleshooting rather than on research. That overhead is manageable for a 200-person biotech with a dedicated informatics team, but it's a significant burden for a small team.
However, there are also many things Benchling genuinely does well, and it's worth pointing these out. It is more flexible and modern than many of the long-established platforms in this space. For molecular biology-heavy teams, the praise in user reviews is consistent and enthusiastic - the sequence analysis tools, plasmid editor, and cloning workflows are highlights that come up again and again. The user community is large and active, documentation is robust, and product updates ship frequently. Benchling has also embraced an AI-first approach, integrating AI across many layers of the platform - so for teams with the resources and appetite for automation and AI-assisted workflows, it can be a strong fit.
But if you're looking for alternatives because Benchling may not be the right fit for your team - whether due to cost, complexity, or other reasons - here are ten options worth evaluating. I've tried to be honest about the strengths and limitations of each, including the one we’ve built.
1. IGOR
Full disclosure: this is our platform, and while I'll do my best to be balanced, some bias is inevitable.
IGOR is a cloud-based electronic lab notebook with integrated LIMS capabilities built specifically for life science research teams. It was designed by lab scientists, not a software company that happened to enter the life sciences market, which means the decisions about what to prioritize, and how things should actually work, came from direct multi-decade bench experience. The result is something that feels immediately familiar to scientists: intuitive to set up, easy to customize without coding, and straightforward enough that most teams are up and running within days or a couple of weeks. Users can adapt the platform to their own workflows through a point-and-click interface, without any need for IT support or vendor involvement.
The core idea is that your experiment records, raw data, SOPs, and inventory should live in the same system and be properly linked - so when you look at a notebook entry, you can see exactly which reagents, samples, and protocols were involved, and when you look at a sample, you can trace every experiment it's touched. That kind of end-to-end connectivity is something labs often try to stitch together across three or four separate tools. With IGOR, it's native.
That ease of use and flexibility doesn't come at the cost of security or compliance though. IGOR is 21 CFR Part 11 compliant and supports GLP workflows, including tamper-proof audit trails and digital signature workflows built in as standard. Access is managed through unique user authentication with role-based permissions, and enforced at both the application level and the database level using row-level security. Data is encrypted in transit and at rest, and backed up across multiple regions, so there's no single point of failure.
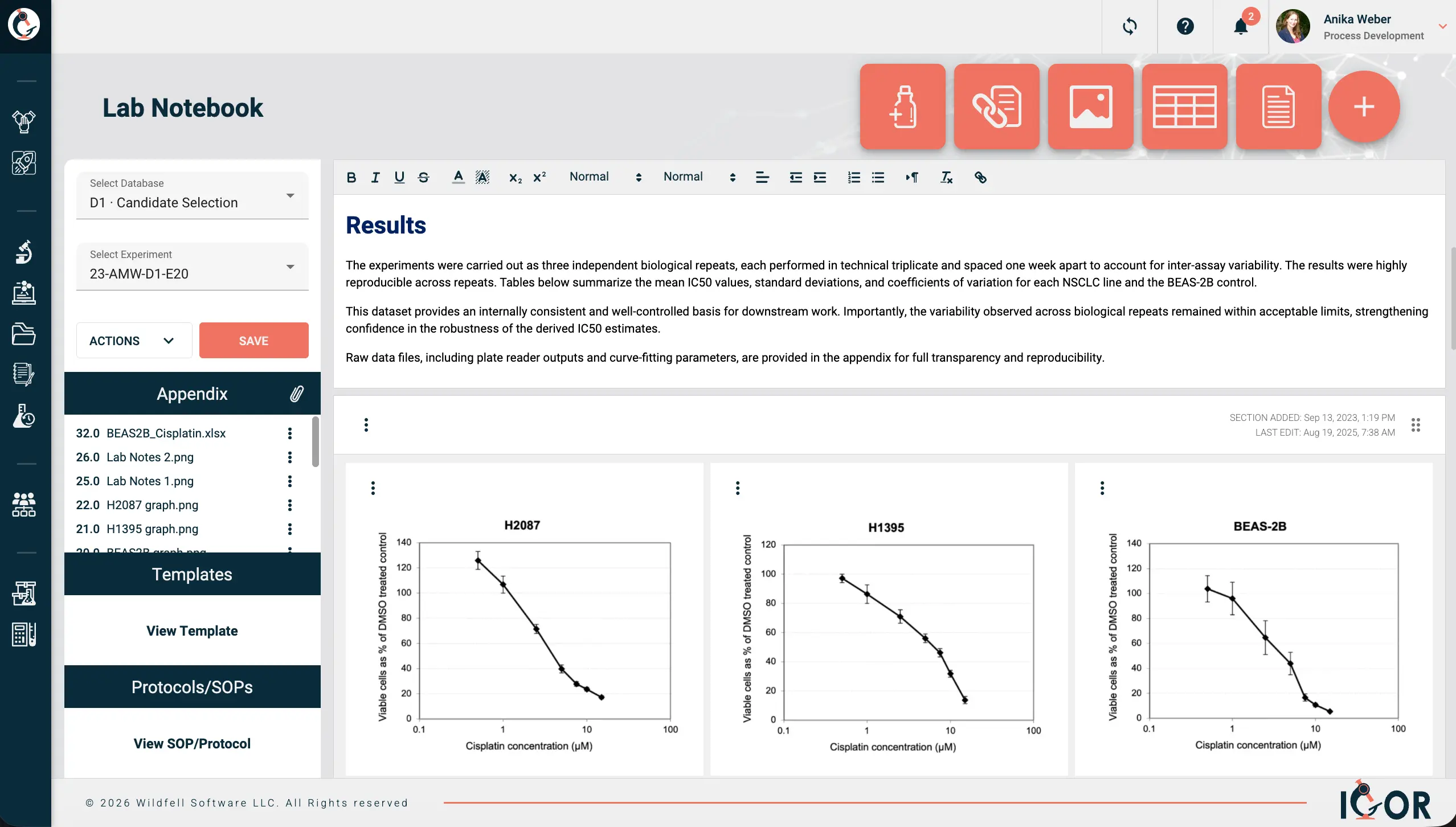
What also sets IGOR apart is the combination of enterprise-level functionality with pricing that's actually accessible to startups and smaller teams - a meaningful differentiator in a market where comprehensive capability typically comes with a price tag to match. You get a genuinely full-featured platform - ELN, LIMS, SOP management, project management, digital signatures, compliance tools - without hidden fees, seat minimums, or surprise add-ons.
What the platform brings together in one integrated system:
- ELN - a rich text editor, image galleries with annotation tools, formula-supported tables with Excel import and export, and direct links to SOPs, samples, reagents, and other materials from your inventory. Any data file type can be attached and linked directly to your entries, so the evidence behind a result stays with the result.
- Lab inventory management with LIMS capabilities - interactive storage maps, sample relationship tracking with visual lineage mapping, full sample history with audit trail, and real-time inventory visibility across your team.
- SOP management - write, version, and approve procedures in the same system where your team actually works. No more hunting through shared drives for the current version, or finding out someone followed an outdated protocol three weeks after the fact.
- Experiment templates - fully customizable and shareable across teams, so common assays are set up consistently from the start rather than rebuilt from memory each time.
- Project management - built-in project tracking to coordinate research activities across teams and departments, with direct links to the experimental data generated within each project.
- Real-time research collaboration - secure data sharing and digital signature workflows for review and approval, so sign-off doesn't mean printing something out and tracking down a PI.
- Fine-grained user permissions and access controls - configurable at the team workspace level, so access reflects how your lab is actually structured.
- Dedicated Team Workspaces - independently configurable per group, department, or research project. Your cell biology team and your chemistry team each get a setup that works for them, without one cluttering the other.
- Companion ELN mobile app - lets bench scientists photograph resultsor notes and upload observations directly to the right notebook entry from the bench.
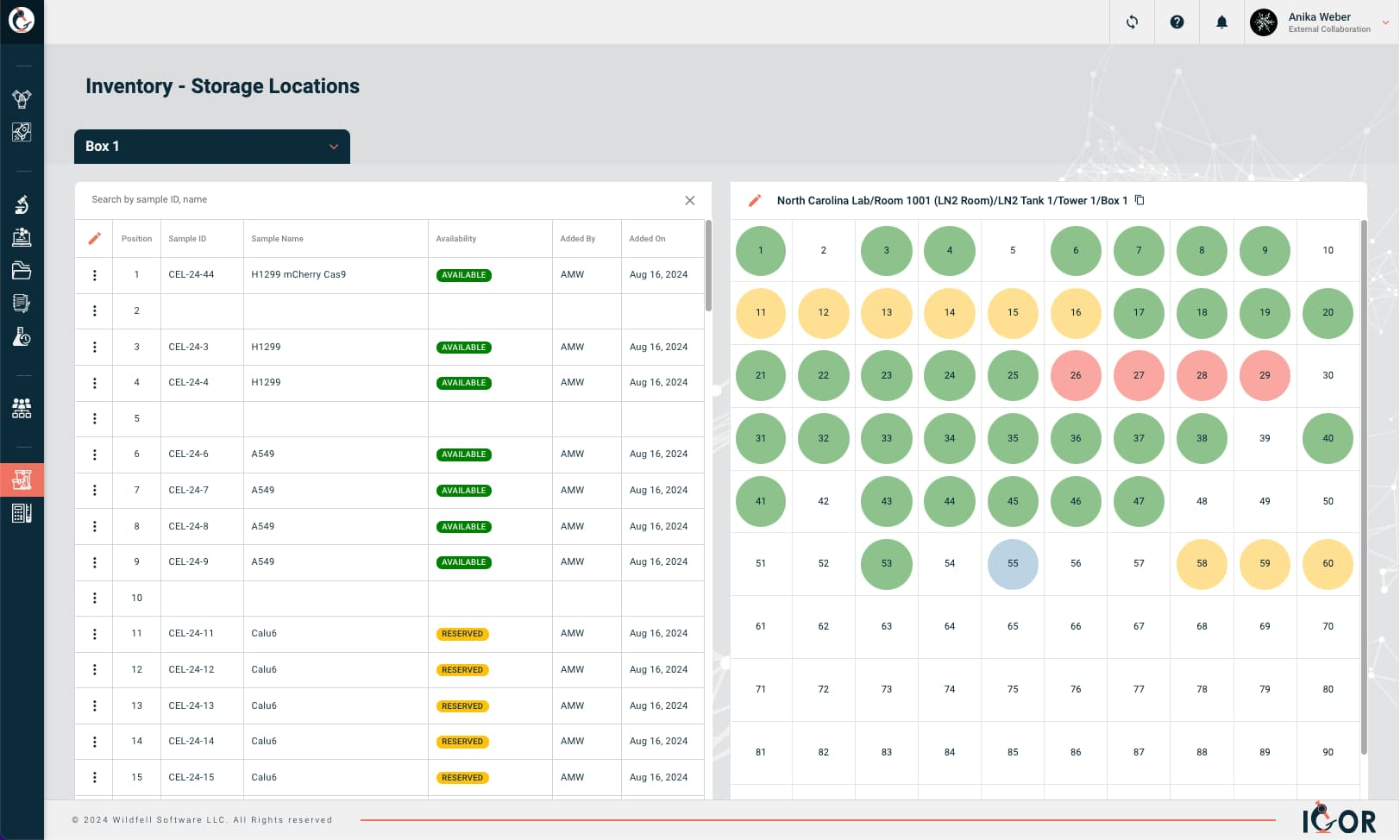
What users say (G2 and Capterra): Reviews are consistently positive. Ease of use and minimal setup time come up repeatedly - multiple reviewers highlight how quickly teams get started. Customer support is mentioned in nearly every review as a standout strength. The combination of ELN and inventory in one connected system is frequently cited as a genuine time-saver, and the template and compliance features get specific praise from regulated lab environments. On the critical side, some users have asked for more self-serve training materials and how-to videos. It's fair feedback, and we've acted on it: we just published an IGOR User Handbook that covers how to use all functionalities of IGOR in detail. We've also been extending our knowledge base considerably and are currently adding a library of self-serve training videos available to all IGOR customers.
Where we're still building: IGOR doesn't yet have dedicated chemistry or molecular biology tools - if your team lives in plasmid design and sequence analysis, Benchling still has an edge there. Instrument integration for lab automation and AI-powered data summaries and visualizations are both on the roadmap. We're a newer platform, which means the community is smaller and you won't find as many third-party tutorials about it. We're growing fast, but we're not pretending to be at Benchling's scale yet.
Best for: Small to mid-size research labs in biotech, pharma, and academia that need a fully integrated ELN and LIMS without a steep learning curve or a steep price tag. Especially well suited for teams that found Benchling too expensive, too complex, or too rigid to adapt across different research groups. In my opinion IGOR is the best integrated lab data management platform for startups, though I'll admit some bias here - I genuinely love what we've built, and I often wish I'd had it when I was still working at the bench.
2. Cenevo (formerly Labguru)
If you've evaluated Labguru before, it's worth knowing the platform has gone through a significant change. In 2024, Labguru was acquired by Titian Software as part of a Battery Ventures life sciences portfolio. In July 2025, the two companies rebranded as Cenevo - combining Labguru's experimental-centric ELN, LIMS, and inventory management capabilities with Titian's Mosaic platform, which specializes in sample-centric lab operations and automation connectivity with over 150 device and system integrations.
The combined entity serves over 950 customers across academic, biotech, and pharma environments - including eight of the top ten pharma companies. The platform now positions itself around the concept of the "smart connected lab," with an emphasis on automation, AI readiness, and full data lifecycle management from research through production.
What users say (G2 and Capterra): Labguru holds a 4.6/5 on G2. Users consistently praise the inventory management, audit trail functionality, and the ability to link experiments to samples and reagents. Customizability is a recurring positive - teams appreciate building out workflows without heavy IT involvement, and customer support is frequently called out as a strength. On the negative side, the most common complaints are around speed, particularly during bulk actions and with larger datasets. Table functionality is frequently flagged as limited, with poor copy-paste behavior from Excel. Some reviewers also note that onboarding new team members requires more effort than expected due to the volume of navigation steps involved.
The rebrand is recent enough that some aspects of the combined Cenovo product experience are still evolving. Whether the UX criticisms that followed Labguru have been meaningfully addressed is something worth pressure-testing during a trial.
Best for: Mid-to-large labs that need a combined ELN and LIMS with strong automation connectivity and don't mind enterprise pricing and onboarding complexity. Good for organizations already in the Battery Ventures ecosystem or with existing relationships with either Labguru or Titian.
3. SciNote
SciNote is an open-source ELN with a strong following in academic and government research settings. The base product is free for individual users, and pricing across its paid tiers is generally more accessible than many alternatives - making it a natural first stop for budget-constrained labs. It handles the fundamentals of experiment documentation well: protocol management, team collaboration, file attachments, and basic inventory tracking.
For regulated environments, SciNote offers a premium tier with 21 CFR Part 11 compliance features and GxP support. The interface is clean and intuitive, and most users report that getting started requires minimal ramp-up time.
The trade-offs are in what SciNote doesn't cover. Advanced automation is limited. The LIMS functionality is basic compared to dedicated LIMS platforms. AI-driven analytics and workflow features are largely absent. And because the foundation is open-source, support and update consistency can vary depending on which version you're running.
What users say (G2 and Capterra): On the positive side, users consistently highlight the customer support team as responsive and helpful, and the automatic report generation and template system get specific praise for saving time on repetitive documentation. The ability to centralize data and eliminate paper notebooks is also frequently mentioned as a genuine step forward for labs making the transition for the first time. On the critical side, navigation is a recurring complaint - the projects/experiments/tasks hierarchy feels unintuitive to many users, and finding files takes more clicks than it should. Several reviewers describe the interface as clunky, with redundant steps and a layout that doesn't always behave as expected. Integration with external data analysis tools is limited, which becomes a real problem for teams working with larger datasets. The results and data management features are frequently flagged as insufficiently robust for anything beyond basic documentation needs.
Labs that start on SciNote and scale to multi-team or multi-department workflows often find they've outgrown it - which means a migration to something more capable, and the disruption that comes with it.
Best for: Academic labs, government research, and early-stage startups with tight budgets that need a reliable, no-frills ELN to get started.
4. Sapio Sciences
Sapio sits at the opposite end of the spectrum from SciNote. It's an enterprise-grade platform combining LIMS, ELN, and what they call a "Scientific Data Cloud" into a configurable system aimed at large pharma, genomics, and clinical labs. They've also launched Sapio ELaiN, an AI-assisted notebook that actively supports experiment design and analysis rather than just serving as a data repository.
Sapio's strength is handling complex, high-throughput, data-heavy environments: NGS workflows, bioanalysis, stability studies, histopathology. The platform is highly configurable via a no-code/low-code builder, and they put significant emphasis on making sure data is structured and AI-ready at the point of capture.
What users say (G2): Sapio has a relatively small review base, but the themes are consistent. Users appreciate the configurability, the out-of-the-box workflow templates, and the in vivo study design tools. The support team is described as engaged and willing to work through implementation challenges. On the negative side, documentation has historically been a significant gap - reviewers frequently note that specific features lack adequate explanation, requiring support contact for basic how-to questions. The learning curve is steep, particularly for ELN and SDMS components, and implementation typically takes longer than expected.
Pricing reflects the enterprise market, which puts it out of reach for most smaller organizations. If you're a 15-person startup, Sapio is probably more platform than you need.
Best for: Large pharma, genomics companies, and clinical labs with complex workflows, high data volumes, and the IT infrastructure to support a sophisticated implementation.
5. SciSure (formerly eLabNext / eLabJournal)
SciSure came together through the merger of eLabNext - the company behind the widely used eLabJournal ELN - and SciShield, an Environmental Health & Safety platform. The resulting product is what they call a Scientific Management Platform (SMP): a unified environment that combines ELN and inventory management with EHS, compliance, training, and safety documentation.
eLabJournal had a strong following, particularly in European academic labs and research institutions, and the underlying ELN is well regarded for being genuinely flexible and straightforward to use. Version control, protocol management, barcode scanning, and a mobile app are all solid. The platform integrates with a range of third-party tools through an API and a marketplace of add-ons.
What users say (G2 and Capterra): The dominant theme in positive reviews is ease of use and customer support - SciSure regularly wins "Ease of Use" badges on Capterra, and the onboarding team is frequently praised. Users particularly like the inventory module, sample-experiment linking, and audit trail functionality. The Project > Study > Experiment structure is noted as helping labs enforce documentation discipline. On the critical side, file management is the most consistent frustration: the platform doesn't allow selecting and uploading multiple files simultaneously, and doesn't remember which folder a user was in during repeated uploads. Some advanced features have a steeper learning curve, and the EHS/SciShield components carry additional cost that smaller organizations can struggle to justify. A few users flag that the Excel online integration doesn't fully replicate regular Excel behavior.
One area worth probing during evaluation is the depth of LIMS functionality. The sample and inventory management is solid for most research workflows, but labs with highly complex sample genealogy requirements or heavy QC needs may find it less comprehensive than dedicated LIMS platforms.
Best for: Research institutions, biotech companies, and academic labs - particularly those in environments where EHS and compliance documentation are meaningful operational considerations, not just an occasional audit concern.
6. LabArchives
LabArchives has carved out a strong position in academic and government research by doing a few things very well. Their partnership strategy - including integrations with university systems and FedRAMP authorization for federal labs - gives them institutional credibility that's genuinely hard to match. In 2021 LabArchives was acquired by Insightful Science and then integrated into the broader Dotmatics platform in 2022. The platform now holds FedRAMP Moderate authorization and has been adopted across tens of thousands of NIH researchers. If your university already has a LabArchives site license, it's often the path of least resistance.
The ELN functionality is solid for documentation, collaboration, and data sharing. Version control and audit trails are well-implemented. And because LabArchives has been focused on the academic market for years, the product reflects quite well how academic labs actually work.
What users say (G2, Capterra, ResearchGate): Positive reviews tend to come from academic users who appreciate the price point, the university integrations, and the core documentation functionality. On the critical side, the interface is consistently described as outdated and less intuitive than modern alternatives. Users on ResearchGate and other forums have flagged specific frustrations including aggressive auto-logout during experiments, buggy rich text editing, and poor copy-paste behavior from Word documents. Support responsiveness has been cited as a concern. Limited mobile functionality comes up regularly. These are long-standing complaints rather than new ones, suggesting the platform has not fully kept pace with UX expectations in the broader market.
The more significant structural limitation is that LabArchives is fundamentally an ELN. Its inventory capabilities are frequently cited in reviews as too limited to be genuinely useful, and there is no meaningful LIMS functionality to speak of. For academic labs doing basic research, that's usually fine. For labs growing into more structured workflows - or those that straddle academia and industry - it becomes an issue fairly quickly.
Best for: Academic and government research labs, especially those at institutions with existing LabArchives agreements.
7. LabWare LIMS
LabWare is one of the most established names in laboratory informatics, with over 30 years in the market and more than 3,000 customers worldwide - including NIH, USDA, GSK, and Pfizer. Its core product is an enterprise-grade LIMS built for high-throughput, compliance-heavy environments across pharmaceuticals, biobanking, bioanalysis, environmental testing, food and beverage, and clinical research. The platform is highly configurable, supports multiple deployment options (cloud or on-premise), and has deep instrument integration capabilities.
LabWare does offer an ELN module (LabWare ELN) as part of what they call their Enterprise Laboratory Platform (ELP). It's not a standalone add-on but a fully integrated component that shares data bidirectionally with the LIMS, meaning experiment records, sample data, reagent inventory, and instrument results all connect within the same system. The ELN supports guided execution of standardized testing methods, instrument data capture, version-controlled method templates, and 21 CFR Part 11 and GLP compliance - making it well suited to regulated quality and manufacturing environments rather than exploratory R&D.
What users say (G2 and Capterra): LabWare's strengths in user reviews are consistent: deep customizability, robust compliance infrastructure, and strong instrument connectivity. Enterprise reviewers on G2 frequently recommend it for complex, multi-site operations. On the critical side, the user interface is a recurring complaint - reviews describe it as dated and not particularly intuitive. The learning curve is steep, configuration can be time-consuming, and integration with some third-party instrument software requires significant effort. Labs with limited IT resources consistently find it difficult to get full value from the platform without substantial investment in setup and training. And of course the price tag - it’s hugely expensive.
Best for: Large enterprises and regulated labs - particularly in pharma, bioanalysis, and industrial QC - that need a proven LIMS with strong compliance credentials and are prepared to invest in implementation. Not the right fit for smaller research labs or teams looking for a quick, low-overhead deployment.
8. CDD Vault
CDD Vault occupies a specific and well-defined niche in this landscape: it's a cloud-based drug discovery informatics platform built around the chemistry-biology interface. If your lab's work centers on compound libraries, structure-activity relationships (SAR), and bioassay data management - particularly in a drug discovery context - CDD Vault covers that ground more purposefully than most general-purpose ELNs.
The platform handles chemical registration, SAR analysis, bioassay data management, inventory, and data visualization in a single environment. There's also an ELN module. Collaboration features are designed around the specific reality of drug discovery teams, including the ability to share selective data with external partners without opening up the full vault.
What users say (G2 and Capterra): CDD Vault reviews are notably positive for a specialized platform. Users in drug discovery consistently praise the intuitive interface for compound and assay data management, the SAR analysis tools, and the selective data-sharing capabilities. The support team - described as being made up of scientists - gets particular credit. On the negative side, the most consistent complaint is around data visualization: for publication-quality figures, users still need to export to GraphPad Prism, as the in-platform graphing doesn't meet that bar. Bulk data uploads can require specific formatting, and some advanced features lack sufficient documentation for new users.
The honest limitation is scope. CDD Vault was built for chemistry-focused drug discovery, and it shows in both the strengths and the gaps. Labs that need broader experiment documentation, non-chemistry workflows, or robust sample tracking outside a compound registry context will find themselves working around its constraints.
Best for: Chemistry-focused drug discovery teams that need rigorous compound data management and SAR analysis. Not the right fit for labs with broad or mixed research modalities.
9. STARLIMS / Labstep
STARLIMS is a long-established enterprise LIMS platform - originally founded in 1986, acquired by Abbott in 2009, and spun out independently in 2021 under private equity ownership - found in over 2,000 laboratories across pharmaceuticals, biotech, CDMOs, food and beverage, public health, and clinical diagnostics. In 2023, STARLIMS acquired Labstep - a modern, cloud-based ELN originally popular in academic settings and now expanding into industry - to address a gap in its portfolio around R&D-stage experiment documentation.
The two products serve meaningfully different parts of the lab workflow. STARLIMS is the enterprise LIMS: powerful, compliance-ready, and built for regulated quality and manufacturing environments with deep roots in FDA, ISO, GxP, and HIPAA-regulated contexts. Labstep is the lighter-weight ELN layer: intuitive, quick to deploy, and designed around interactive step-by-step protocols that structure data capture in real time.
What users say (G2 and Capterra): STARLIMS holds a 4.5/5 on G2, with users consistently praising sample lifecycle management, scalability, and compliance infrastructure. Instrument integration and data traceability are highlighted as standout strengths. On the critical side, the SDMS and ELN components of STARLIMS are noted as less polished than the core LIMS - issue resolution within the ELN can be slow, and search functionality and statistical analysis tools are frequently flagged as inadequate. Results entry can slow significantly under heavy data volumes. The learning curve is steep, particularly for new users approaching the SDMS modules. Labstep on its own receives positive feedback for ease of use and quick onboarding, but teams in regulated industries often report outgrowing it relatively quickly as workflows become more complex.
Best for: Organizations already evaluating STARLIMS for their LIMS needs who want a paired ELN, or R&D teams drawn to Labstep's protocol-centric approach and university track record. Independent validation of how it performs across different lab sizes and workflow complexity is worth seeking out during a trial.
10. IDBS (Polar / E-WorkBook)
IDBS is the name you'll encounter most often when talking to informatics teams at large pharma. The company, acquired by Danaher in 2017, has been providing R&D data management software for over 35 years, and their customer base reflects that tenure - around 75% of the top 20 biopharma companies have been customers for over a decade. Their current platform, Polar, combines ELN, LES (Lab Execution System), and LIMS capabilities in a single GxP-compliant environment designed for the full drug development lifecycle.
For large regulated organizations, that depth and track record is genuinely valuable. IDBS handles biologics workflows, process development, bioanalytical data, stability studies, and complex multi-site environments with the kind of rigor that a heavily validated pharma context demands.
What users say (G2 and industry analysis): IDBS doesn't attract the same volume of consumer-facing reviews as smaller platforms - it's not the kind of software that individuals evaluate independently. What does surface though is consistent: the platform scores lower on ease-of-use metrics relative to peers, which is largely expected given its complexity. Users who do review it tend to work at large pharma organizations and describe the platform as powerful but demanding. Customer support has been a friction point for some organizations, with concerns about fees for what might reasonably be considered standard support. Smaller or more agile organizations consistently find the rigidity frustrating, as customizations require coding knowledge or customer support.
The trade-offs are worth being direct about. IDBS is expensive - enterprise pricing with implementation costs that reflect the platform's complexity. It was built for the compliance and governance needs of large pharma, and that DNA is present throughout - which is exactly what you want if you're a large pharma company, and a significant headwind if you're not.
Best for: Large pharmaceutical companies, CROs, and clinical labs operating in highly regulated environments where compliance rigor, established vendor relationships, and long-term platform stability outweigh concerns about cost, flexibility, or implementation speed.
How to Actually Choose
After working through all of these, the decision usually comes down to a few concrete questions. What's your realistic per-user budget per year? How much IT bandwidth do you have for implementation and ongoing maintenance? Is compliance a current requirement, or something you'll need to grow into? And - probably the most important question - will the people who actually use this every day adopt it and stick with it?
My advice, and yes I'm biased because I work on one of these products, is to run trials with two or three options using actual bench scientists. Not a polished vendor demo with someone from sales driving. A real trial where your team enters real data, runs real workflows, and identifies their own friction points. The tool that gets the least resistance from the people who have to open it every morning is probably the right one.
If you're evaluating ELN's for your startup, check out this article on How to choose the best ELN for your Biotech startup.


