IGOR safeguards your research data while supporting compliance with regulatory guidelines. So your team can work with confidence, no matter where science takes you.

We use industry-leading cloud infrastructure and modern security best practices to ensure your data is protected.
IGOR is built to support 21 CFR Part 11 compliance, helping regulated teams manage electronic records and signatures with confidence.


For research organizations in the UK, IGOR supports compliance with the Human Tissue Authority (HTA) by providing full traceability of human tissue samples - from arrival on-site through to disposal. The same rigorous tracking and audit features also support the safe handling, documentation, and disposal of hazardous materials in line with institutional and regulatory requirements.
IGOR supports your SOP-driven workflows and standardized data capture - helping you stay compliant with GLP and GMP expectations. From creating reusable experiment templates to managing SOP approval workflows, IGOR helps you maintain consistent processes and high data standards across your entire organization.


As a cloud-based platform trusted by research teams around the world, IGOR puts data protection and privacy at the core of everything we do. We implement privacy and security best practices that meet the needs of biotech, pharma, and academic teams handling sensitive scientific and personal data.
From the FDA’s GLP, GMP, and 21 CFR Part 11 to the HTA and EMA, the regulatory landscape can feel overwhelming. IGOR helps your team navigate it with confidence.
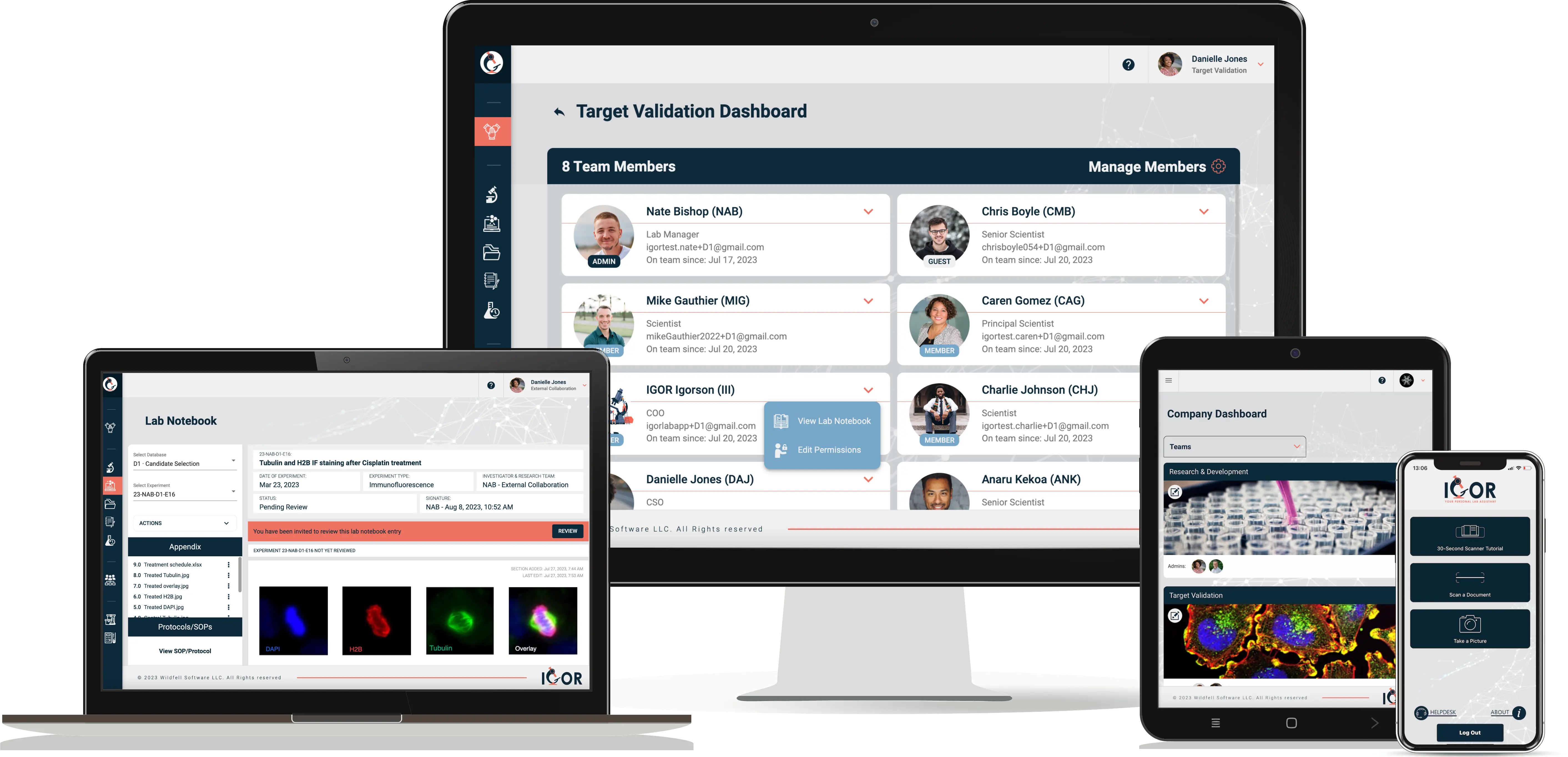
More than just an ELN, IGOR includes built-in safeguards like role-based access, audit trails, digital signatures, and SOP management - plus streamlined workflows that make documentation and approvals part of your everyday process.
Your team is responsible for compliance - but IGOR gives you the tools to make it manageable, reliable, and part of your everyday lab operations.
SOP Management
Create, share, and manage standard operating procedures within your lab workflows - a must for regulated environments.
21 CFR Part 11 Compliance
Digital signatures, audit trails, and access controls to meet FDA requirements for electronic systems.
GLP & GMP Support
Built-in SOP management, experiment templates for standardized data capture, and detailed audit trails support GxP.
Version Control & Data Integrity
Every edit to a notebook entry is logged automatically. Nothing is ever permanently overwritten, and all changes are attributed, timestamped, and audit-ready.
Review & Approval Workflows
Define who authors, witnesses, and approves notebook entries and SOPs. Each signature is timestamped and permanently linked to the record - and once witnessed, entries lock.
GDPR Compliance
IGOR is GDPR compliant and certified under the EU-U.S. Data Privacy Framework. Your organisation retains full data ownership, with subprocessors listed publicly and a DPA available.
Let us show you how easy lab data management can be.
21 CFR Part 11 is an FDA regulation that sets out the requirements for electronic records and electronic signatures in regulated industries; essentially, it's the rules that make a digital record legally equivalent to a paper one. If your lab is in pharma, biotech, or medical devices, and you're creating records that the FDA could ask to inspect, those records need to meet Part 11 standards.
In practice, that means your ELN needs to do a few specific things: restrict access to authorized users only, generate tamper-proof audit trails that log every change with a timestamp and the identity of who made it, support compliant electronic signatures that are unique to each individual and permanently linked to the records they sign, and lock records once they've been reviewed and witnessed so they can't be altered without a traceable correction.
It's worth knowing that no software vendor can make your lab "21 CFR Part 11 compliant" on its own - compliance also depends on how your team uses the system, your SOPs, and how you've validated it for your specific workflows. What IGOR does is give you all the technical building blocks you need: closed system access, fine-grained user permissions and access controls, audit trails, version control, digital signature workflows, and entry locking after witnessing - so your team can focus on the procedural side rather than worrying about whether the software is up to the task.
It's a fair question, and one worth asking any vendor directly rather than taking marketing language at face value.
For IGOR specifically: your data is hosted on AWS (Amazon Web Services) infrastructure in SOC2-certified data centers, encrypted end-to-end, and backed up automatically to geographically separate locations so that a failure in one region doesn't put your data at risk. Access controls are enforced at a granular level throughout the platform: administrators can define precisely which team members can view or modify specific repositories, notebook entries, and files, ensuring that data is compartmentalized according to your organization's structure and security requirements. We also run continuous security monitoring.
The practical upshot is that for most research labs, a well-secured cloud platform is actually safer than local servers, which tend to rely on institutional IT infrastructure that varies enormously in quality and rarely has the same level of redundancy. The bigger risk for most labs isn't the cloud itself - it's weak access controls, shared credentials, or no backup strategy at all. Those are problems a good cloud ELN solves, not creates.
If you have specific security requirements, we're happy to walk through our setup in detail.
Yes. IGOR is GDPR compliant and is also certified under the EU-U.S. Data Privacy Framework (DPF), which governs how personal data is transferred between the EU and the United States.
In practical terms, that means you retain ownership of your data, we're transparent about how it's handled and who it's shared with (our subprocessors are listed publicly), and we implement the technical and organizational measures GDPR requires: encryption, access controls, data minimization, and the ability to respond to data subject requests.
If your institution or legal team needs documentation - a Data Processing Agreement, subprocessor list, or details of our Privacy Policy - these are available on our website or on request.
Your data stays yours. If you decide to leave, you can export your records - notebook entries, protocols, inventory data, files - in standard, human-readable formats. We don't hold your data hostage, and we don't make export difficult.
This matters more than it might seem. Data lock-in is a real issue in the ELN and LIMS space - some vendors make it technically painful to leave, which effectively traps you even if the product stops meeting your needs. Before committing to any platform, it's worth asking specifically how export works, what formats are supported, and whether there are any fees or restrictions around it. For IGOR, the answer is straightforward: your data goes where you go.
The short answer: if your lab creates electronic records in connection with work that is regulated by the FDA, Part 11 applies to you. That covers pharmaceutical and biotech companies, medical device manufacturers, CROs, and any lab doing contract work that feeds into an FDA submission - even if your organization isn't itself a drug or device company. If your data will eventually be used to support a regulatory filing, the records you're generating now need to meet Part 11 standards.
For academic labs with no commercial or regulatory pathway in sight, it likely doesn't apply yet. For most biotech startups and growing research teams though, the honest advice is to get the right infrastructure in place early; retrofitting compliance onto an existing system and dataset is significantly more painful than building it in from the start.
One thing worth understanding is that Part 11 compliance isn't something a software vendor can hand you - it's a combination of the right tools and the right processes. What IGOR provides is the technical foundation: audit trails, electronic signatures, access controls, locked records, and version history. How your team documents and conducts its work accounts for the rest.
If you're not sure whether your specific workflows require Part 11 compliance, it's worth a conversation with your regulatory affairs team or a compliance consultant - but having the technical infrastructure already in place gives you a significant head start.